
Daniel Schwarz, osapiens Expert | 17. March 2026 | Lesezeit 10 min.
Chasing down maintenance records and paper logs before a regulatory inspection is a stress no team needs. GxP compliance software is meant to solve this by making documentation a natural part of the workday.




ENTERPRISE-READY
SAP Certification
ISO 27001
EU Hosting
Entra ID & Okta
Multi-Entity
Table of Contents
- Key Facts
- What Does GxP Mean?
- What Is GxP Compliance Software?
- Why GxP Compliance Is Essential for Pharma and Life Sciences Industries
- Key Features of GxP Compliance Software
- Challenges of GxP Compliance Without Software
- GxP Compliance Automation with the osapiens HUB
- Conclusion: The Foundation for Audit-Ready Pharma Maintenance
- FAQ
Instead of scrambling before audits, pharma and life sciences organizations can rely on digitized, traceable maintenance workflows that keep documentation complete, accurate, and audit-ready at all times. As a purpose-built pharma maintenance software, the osapiens HUB helps teams make that shift, turning audit anxiety into audit readiness with built-in audit trails, mobile protocol execution, and seamless SAP integration.
Key Facts
- GxP compliance is mandatory: Regulatory guidelines covering manufacturing, laboratory, clinical, and distribution practices require life sciences organizations to document and trace every process. Non-compliance triggers EMA warning letters, suspended licenses, and market bans.
- Maintenance is a frequent inspection gap: Calibration records, equipment qualification documents, and maintenance logs are among the most commonly cited deficiencies in regulatory audits.
- The fix is digital: Automated audit trails, electronic signatures, and structured validation workflows eliminate the manual errors that put teams at risk during inspections.
- The osapiens HUB keeps your team inspection-ready by digitalizing pharma maintenance workflows with full SAP integration, built-in audit trails, and mobile-first protocol execution to keep teams inspection-ready.
What Does GxP Mean?
GxP stands for “Good [x] Practice,” where the “G” means Good, the “x” is a placeholder for the specific practice area, and the “P” stands for Practice. Together, these regulatory guidelines ensure that products in the life sciences industry are safe, effective, and manufactured to consistent quality standards. The “x” changes depending on the discipline involved. The relevant practice areas for life sciences organizations are:
- GMP = Good Manufacturing Practice: Covers manufacturing processes, quality control, and production documentation.
- GLP = Good Laboratory Practice: Governs non-clinical laboratory studies and data integrity.
- GCP = Good Clinical Practice: Ensures the ethical and scientific quality of clinical trials.
- GDP = Good Distribution Practice: Regulates product quality during transport, storage, and distribution.
- GEP = Good Engineering Practice: Addresses facility and equipment design, commissioning, and maintenance.
What are the GxP requirements for pharma maintenance? Pharma maintenance teams must document every calibration, inspection, and equipment qualification in a traceable, attributable format. Equipment must follow defined maintenance intervals, and all records must be available for regulatory audits. EU Annex 11 governs electronic records and signatures in GxP environments, requiring that systems are validated and that audit trails cannot be altered or deleted.
GxP-ready maintenance starts here
See how the osapiens HUB supports pharma maintenance teams with structured workflows and audit-ready documentation.
What Is GxP Compliance Software?
GxP compliance software refers to digital solutions that help organizations meet these regulatory requirements with less effort and greater accuracy. These tools range from eQMS platforms for document control to CMMS solutions like osapiens HUB, built specifically for regulated environments.
For maintenance teams, GxP compliance software ensures that every work order, calibration event, and equipment qualification is captured with a complete audit trail, providing the documented proof inspectors expect.
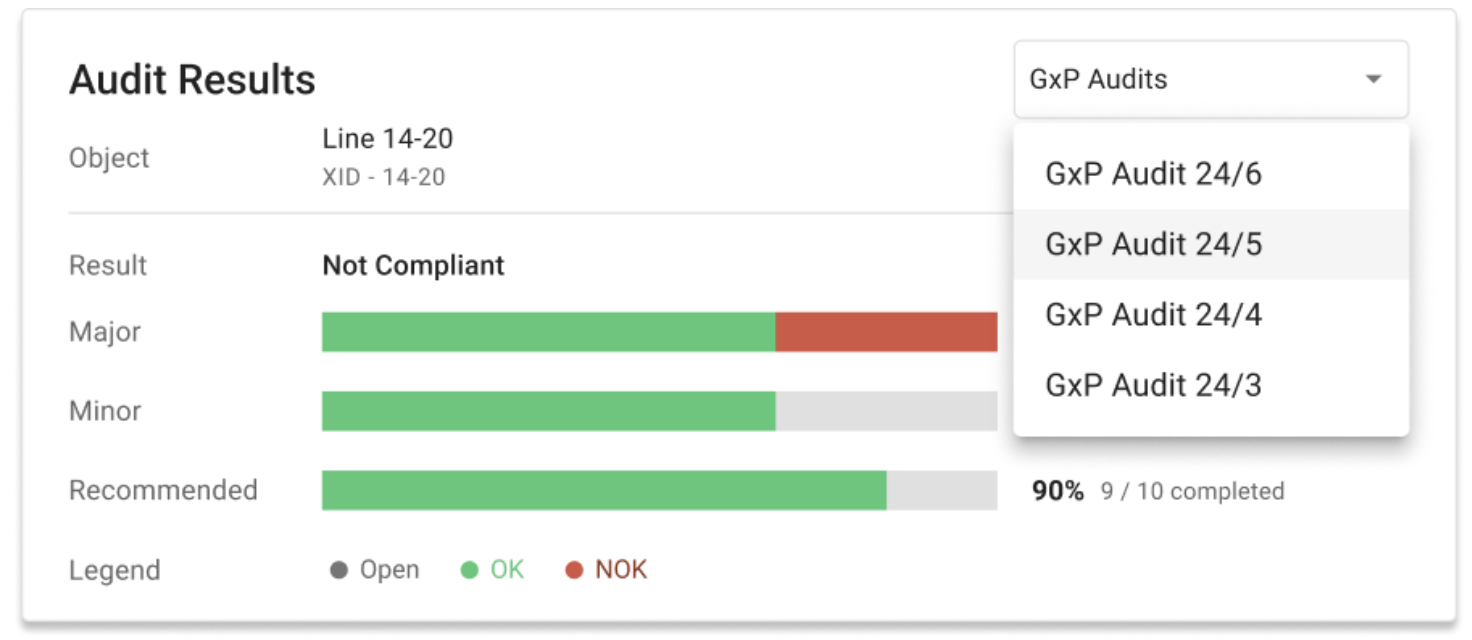
osapiens HUB for Maintenance turns these regulatory requirements into practical, day-to-day workflows, so maintenance teams spend less time chasing records and more time keeping equipment running.
Why GxP Compliance Is Essential for Pharma and Life Sciences Industries
GxP guidelines exist for one fundamental reason: to protect patients. The global Thalidomide crisis between 1960 and 1962, when more than 10,000 babies were born with severe deformities, was a turning point that made good manufacturing practice and good clinical practice mandatory worldwide. Today, these quality standards ensure that every pharmaceutical product reaching patients is safe, effective, and consistently produced.
Non-compliance triggers real consequences that can bring operations to a standstill:
- Warning letters and consent decrees
- Product recalls and delayed batch releases
- Suspended manufacturing licenses
- Market bans across entire regions
Beyond penalties, GxP compliance builds operational trust with regulators like the EMA, national competent authorities, and ICH-aligned agencies worldwide, including the FDA.
For maintenance teams, missing calibration certificates, outdated maintenance intervals, and incomplete equipment logs are among the most frequently cited issues in GxP inspections.
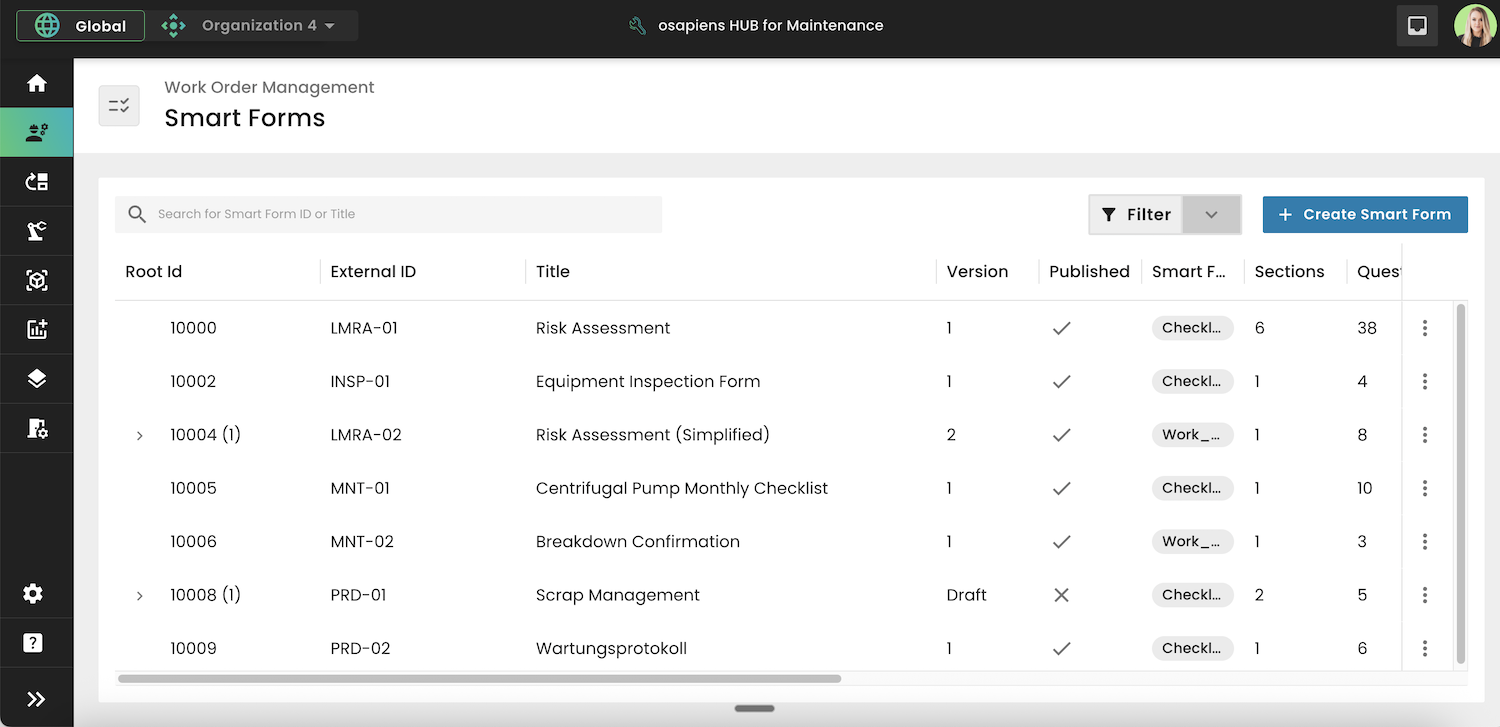
Key Features of GxP Compliance Software
Effective GxP compliance software shares a set of core capabilities that pharma maintenance and quality teams should treat as mandatory. Three features stand out as essential for any regulated environment.
Automated Protocol Execution
GxP software replaces paper-based checklists and manual SOPs with structured digital workflows. Technicians follow guided steps on mobile devices for inspections, calibrations, and safety checks, while documentation is captured automatically as they work. This eliminates the risk of missed steps and significantly reduces administrative burden, giving teams more time to focus on critical maintenance tasks.
Audit Trails and Data Integrity
Immutable audit logs record every action: who did what, when, and why. This aligns with ALCOA+ principles (Attributable, Legible, Contemporaneous, Original, Accurate, Complete, Consistent, Enduring, Available) and is essential for meeting EU Annex 11 and FDA 21 CFR Part 11 requirements. Electronic signatures add another layer of accountability, ensuring only authorized individuals can approve or complete records.
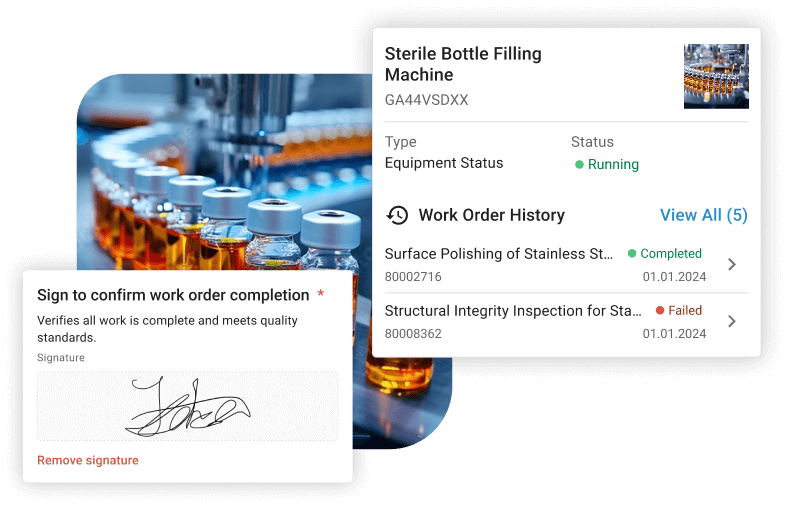
Ongoing Validation Workflows
GxP-compliant systems must be validated not just at implementation but continuously. This involves IQ/OQ/PQ qualification stages throughout the software lifecycle. The industry is shifting from traditional CSV to CSA (Computer Software Assurance), a more risk-based approach. Cloud-based SaaS platforms like osapiens can simplify revalidation cycles compared to on-premise systems, reducing the effort required to maintain a validated state. This makes the osapiens HUB a validated CMMS option for pharma teams that need continuous compliance without the overhead of traditional on-premise validation cycles.
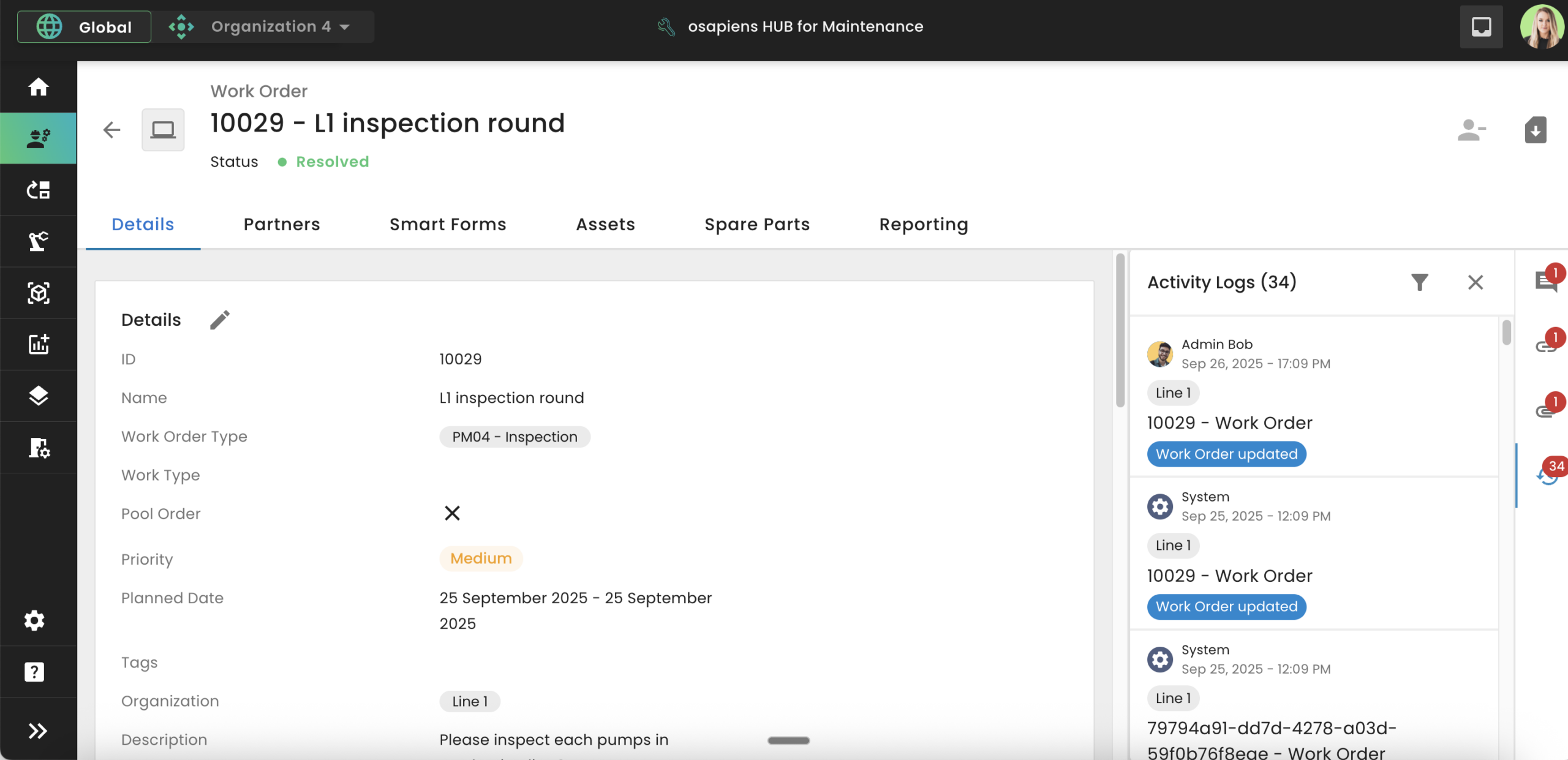
Challenges of GxP Compliance Without Software
Many pharma maintenance teams still rely on Excel spreadsheets, paper-based logs, and disconnected systems to manage GxP documentation. While these methods may work for very small equipment portfolios, they quickly become a liability as operations grow and regulatory scrutiny intensifies. The most common pain points include:
- Incomplete or inconsistent records that fail to meet ALCOA+ data integrity standards, leaving gaps auditors will flag
- Missing calibration certificates that only surface during inspections, when it is too late to fix them
- No traceability for who performed a maintenance task, when it was completed, or what was found
- Media breaks between field work and documentation, where manual transcription introduces errors and delays
- Inability to scale compliance processes as equipment portfolios expand across sites and production lines
Each gap can trigger warning letters, delayed batch releases, or even production shutdowns. The cost of non-compliance, measured in delayed batch releases, suspended manufacturing licenses, and market bans, consistently exceeds the investment in proper GxP compliance software like osapiens HUB for Maintenance.
Stop managing compliance manually
Replace paper checklists and Excel logs with digital, audit-ready maintenance workflows. The osapiens HUB makes GxP compliance operational, from the shop floor to the audit room.
GxP Compliance Automation with the osapiens HUB
Meeting GxP requirements should not mean more paperwork for your maintenance team. The osapiens HUB for Maintenance bridges the gap between strict regulatory guidelines and daily operations by digitizing every compliance-critical workflow, from calibration scheduling to audit trail documentation.
| GxP Requirement | How the osapiens HUB Delivers |
|---|---|
| Equipment inventory with maintenance records | Centralized digital asset maintenance software with complete maintenance history |
| Documented calibration and maintenance schedules | Automated scheduling with reminders and escalation workflows |
| Traceable logs (time, date, personnel, findings) | Immutable audit trails with electronic signatures via mobile app |
| Continuous equipment health monitoring | Real-time dashboards and reporting integrated with SAP PM |
| Validation and qualification documentation | Digital IQ/OQ/PQ workflow support with version-controlled records |
What sets the osapiens HUB apart is its SAP-certified integration, ensuring that maintenance data flows seamlessly between shop-floor execution and enterprise systems without manual transfers. The mobile-first design lets technicians complete inspections, capture electronic signatures, and close work orders directly on the production floor. And because the platform scales from a single site to multi-plant operations, enterprise organizations across regulated industries already rely on it to simplify compliance across growing equipment portfolios. 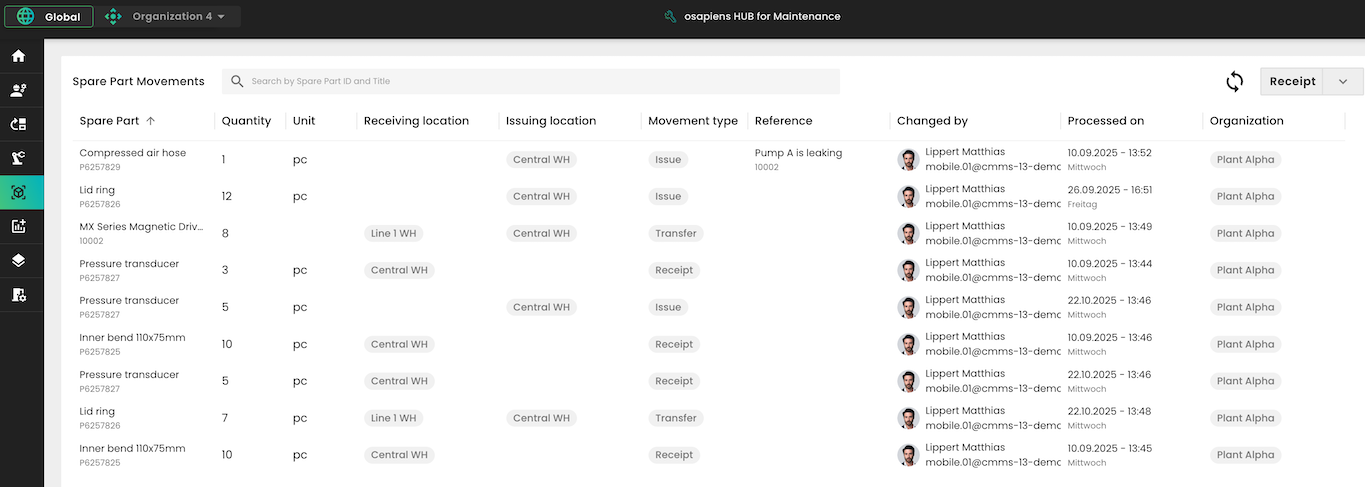
Here is what sets the osapiens HUB apart for GxP-regulated maintenance teams:
- SAP-certified integration: Maintenance data flows seamlessly between shop-floor execution and SAP PM, without manual transfers and eliminating data silos and duplicate entry.
- Mobile-first execution: Technicians complete inspections, capture electronic signatures, and close work orders directly on the production floor, even offline.
- Automated preventive maintenance: Calibration and qualification workflows are scheduled and triggered automatically, based on time intervals or usage, keeping equipment continuously compliant.
- Spare parts traceability: Every part used in a maintenance task is logged and traceable. This is critical for GxP environments where material provenance must be documented.
- Scalable from one site to many: Whether you operate a single production facility or a multi-plant network, the osapiens HUB grows with your compliance requirements.
- Real-time dashboards: Overdue calibrations, open work orders, and compliance status across all assets are visible at a glance and exportable for audits.
Conclusion: The Foundation for Audit-Ready Pharma Maintenance
GxP compliance in pharma is not optional, and the complexity of regulatory requirements makes the right software essential. Manual processes create risk. The right GxP compliance software transforms maintenance from a compliance liability into a competitive advantage, ensuring every calibration, inspection, and work order is documented, traceable, and audit-ready.
The osapiens HUB for Maintenance was built for exactly this challenge. It combines enterprise-grade compliance features with an intuitive, mobile-first experience that technicians actually want to use, backed by SAP-certified integration for seamless data flow.
Stop Risking Audit Failures. Start Documenting Right
Missing records, manual workarounds, and paper-based logs put your GxP compliance at risk every single day. The osapiens HUB gives your team traceable, audit-ready documentation – automatically.FAQ
How does GxP compliance software work?
GxP compliance software replaces manual, paper-based processes with structured digital workflows. When a technician performs a maintenance task, the software guides them step by step, captures every action with a timestamp and user attribution, and stores the record in an immutable audit log. The result: complete, traceable GxP documentation that is available for inspection at any time, without additional administrative effort.
Which ERP software is best for GxP compliance?
No single ERP is universally “best.” SAP is the most widely used in pharma, but GxP compliance depends on proper validation against EU Annex 11 and, for internationally active organisations, FDA 21 CFR Part 11. Layering specialized tools like a CMMS on top is essential for maintenance-specific regulatory requirements.
What happens if you fail a GxP inspection?
A failed GxP inspection can trigger warning letters, forced product recalls, suspended manufacturing licenses, or market bans across EU member states. In serious cases, it can lead to production shutdowns while corrective actions are implemented. The most common root causes are incomplete maintenance records, missing calibration certificates, and lack of traceability. All this can be prevented with a validated CMMS like the osapiens HUB.
How much does GxP compliance software cost per user or per site?
Costs vary widely based on scope, user count, and integration complexity. Specialized tools can start around thousands of euros per year, while enterprise platforms scale higher. The osapiens HUB offers a freemium model so teams can start without upfront investment and scale as needed.
How does the osapiens HUB support GxP compliance in pharma maintenance?
The osapiens HUB digitizes maintenance workflows with built-in audit trails, electronic signatures, calibration scheduling, and mobile protocol execution. Every maintenance event is documented, traceable, and aligned with GxP standards like 21 CFR Part 11.
Does the osapiens HUB integrate with SAP in pharma environments?
Yes. The osapiens HUB is SAP-certified and integrates seamlessly with SAP PM. Maintenance data such as work orders, equipment records, and calibration logs syncs bidirectionally, eliminating manual data transfer and ensuring a single source of truth for regulated operations.
Start using osapiens for free
Improve your uptime, extend the life of your equipment, and simplify work order management.
Related content

Bending Machine Maintenance Checklist – Tasks, Safety & Digital Execution

Booster Pump Maintenance Checklist – Tasks, Tips & Template

Laboratory Equipment Maintenance Checklist – Tasks, Best Practices & Digital Solution

Roofing Maintenance Checklist – Tasks, Tips & Best Practices

Granulation Equipment Maintenance Checklist – Tasks, Tips & Best Practices

Air Compressor Maintenance Checklist – Tasks, Tips & Template

Elevator Preventive Maintenance Checklist – Tasks, Safety & Digital Execution

What is a CMMS? A Guide to Modern Maintenance Software

Unlocking Operational Excellence Through Preventive Maintenance

Assembly Line Equipment Maintenance Checklist – Tasks, Tips & Digital Execution

Automated Guided Vehicle Maintenance Checklist – Tasks, Safety & Digital Execution












